Lewis Structure
The electronegativity of hydrogen is 2.20, of carbon is 2.55, and of oxygen is 3.44. Therefore, the bond nature between carbon and oxygen is 2.55 subtracted from 3.44, which equals 0.89. This is a moderately covalent bond. The bond nature between carbon and hydrogen is 2.20 subtracted from 2.55, which equals 0.35. This is a very covalent bond.
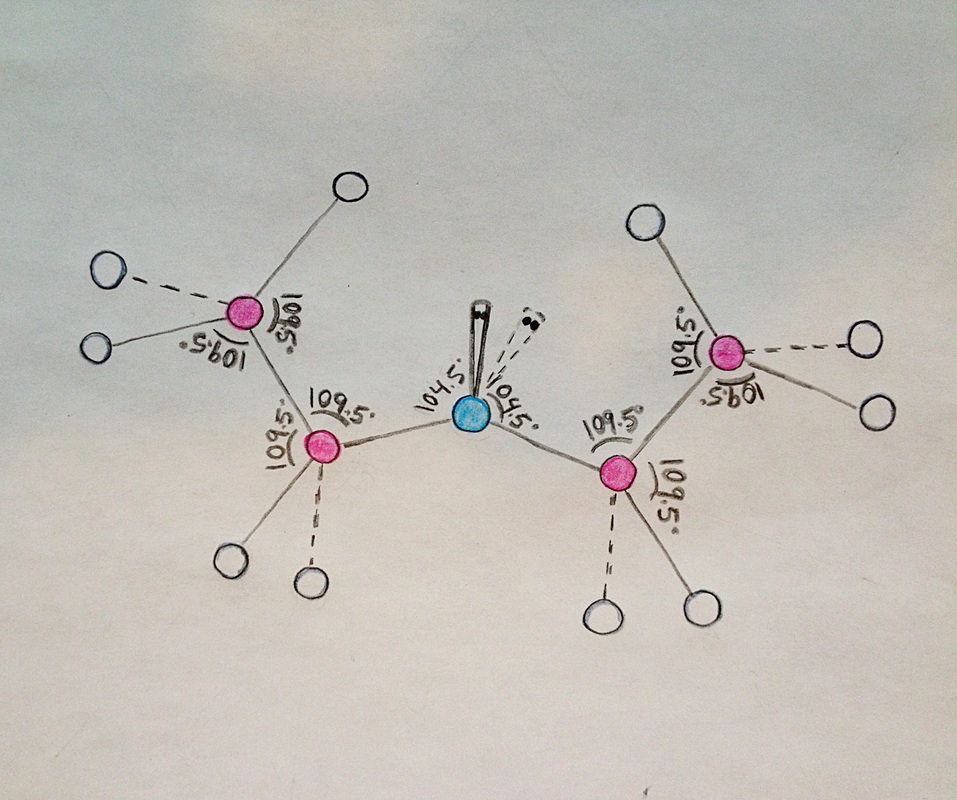
Molecule Drawing
Color Key:
Blue= Oxygen
Pink= Carbon
White= Hydrogen
Grey= Unpaired Electrons
(The unpaired electrons are located above the oxygen atom.)
Diethyl ether is formed by four tetrahedron (AX4) and one bent shape (AX2E2). The oxygen bonded with two carbon atoms and containing two unpaired electrons forms a bent shape. The bond angles in a bent shape are 104.5 degrees. There are two tetrahedron on each side of the oxygen molecule. One consists of carbon bonded with one oxygen atom, two hydrogen atoms, and one carbon atom. The other consist of carbon bonded with three hydrogen atoms and one carbon atom. The bond angles in tetrahedron are 109.5 degrees.
Blue= Oxygen
Pink= Carbon
White= Hydrogen
Grey= Unpaired Electrons
(The unpaired electrons are located above the oxygen atom.)
Diethyl ether is formed by four tetrahedron (AX4) and one bent shape (AX2E2). The oxygen bonded with two carbon atoms and containing two unpaired electrons forms a bent shape. The bond angles in a bent shape are 104.5 degrees. There are two tetrahedron on each side of the oxygen molecule. One consists of carbon bonded with one oxygen atom, two hydrogen atoms, and one carbon atom. The other consist of carbon bonded with three hydrogen atoms and one carbon atom. The bond angles in tetrahedron are 109.5 degrees.
Intermolecular Forces
The polarity of diethyl ether is debated. Solvents with a dielectric constant above five are often said to be polar, and solvents with a dielectric constant below five are often said to be non-polar. Diethyl ether's dielectric constant is 4.3, so some consider it to be non-polar. However, others argue that diethyl ether is polar due to the geometry of the molecule that does not cause the oxygen-carbon bond dipoles to cancel. In other words, the electrons are distributed unevenly, which causes high and low areas of electron density. The unshared electrons above the oxygen result in a more negative charge than other regions of the molecule. Therefore, diethyl ether can be classified as weakly polar.
The types of intermolecular forces include dipole-dipole, hydrogen-bonding, and dispersion. Dipole-dipole occurs between two polar molecules when the more positively charged end of one molecule is attracted to the more negatively charged end of another molecule. Hydrogen-bonding can occur when the hydrogen atom of one molecule is attracted to the oxygen, nitrogen, or fluorine atom of another. Dispersion can occur between any two molecules in a liquid or solid. The electrons shift to create a necessary charge for attraction, which creates a temporary dipole of a non-polar molecule. Dispersion is the weakest form of attraction.
The types of intermolecular forces include dipole-dipole, hydrogen-bonding, and dispersion. Dipole-dipole occurs between two polar molecules when the more positively charged end of one molecule is attracted to the more negatively charged end of another molecule. Hydrogen-bonding can occur when the hydrogen atom of one molecule is attracted to the oxygen, nitrogen, or fluorine atom of another. Dispersion can occur between any two molecules in a liquid or solid. The electrons shift to create a necessary charge for attraction, which creates a temporary dipole of a non-polar molecule. Dispersion is the weakest form of attraction.
|
. The image to the left shows a possible hydrogen bond formed by two diethyl ether molecules. The hydrogen of one molecule is attracted to the oxygen of the other.
In addition, this image shows a possible dipole-dipole attraction, because the negative unpaired electrons of one molecule are attracted to a more positively charged area of another molecule. Since dispersion can occur between any two molecules, the force of attraction of dispersion would be present between two diethyl ether molecules. |